Products are for professional/laboratory use only.
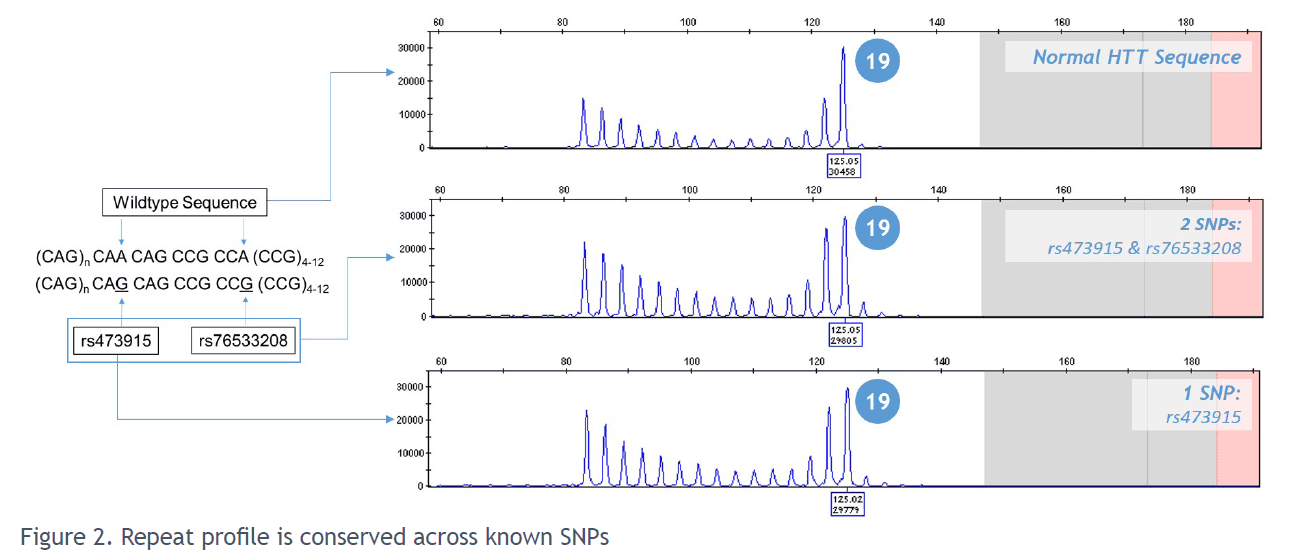
The AmplideX® PCR/CE HTT kit simplifies the detection and sizing of the CAG trinucleotide repeats in the HTT gene by overcoming the challenges of adjacent SNPs and variable CAG repeats which can complicate primer binding and cause allele dropouts.
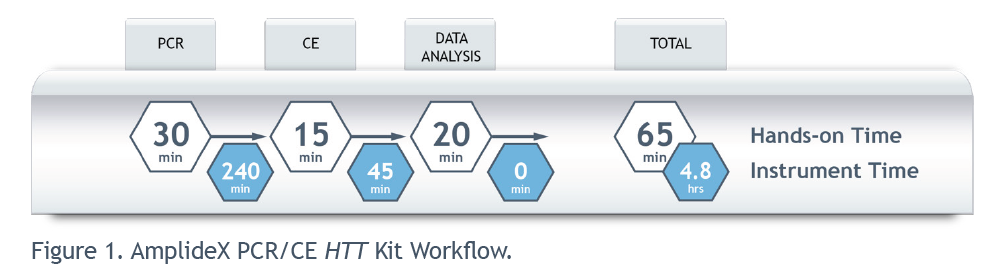
The assay, which is optimised for use on widely established CE systems and requires minimal hands-on time, provides an easy-to-implement solution for laboratories to analyse the CAG repeats in the HTT gene without the need for multiple PCRs.
Benefits of AmplideX® PCR/CE HTT Kit include:
- Reduced complexity: a PCR solution for GC-rich amplification and detection which eliminates the need for multiple PCR resulting in a straight forward analysis
- Quality results: the unique two-primer design provide reliable, unambiguous results and a robust stutter peak pattern
- Similar workflow to AmplideX® FMR1 and DMPK assays to ease implementation and testing
- All-inclusive kit which includes all reagents necessary to confirm presence of CAG expansions in HTT
Reliable, unambiguous results and a robust stutter peak pattern
*Research Use Only.
Read the launch announcement from Asuragen
View more information about this kit
Contact us for a quote