Products are for professional/laboratory use only.
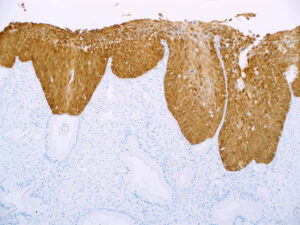
p16INK4A (also known as 16P04) IHC is the most common test run on cervical biopsies to determine the presence of cervical intraepithelial neoplasms (CIN) caused by HPV and is considered a surrogate test for HPV testing in squamous intraepithelial neoplasms.
Inclusion of p16 in the panel of IHC markers run on biopsies is essential to determine whether CIN is present and what may be the severity of the neoplasm.
- Nuclear and cytoplasmic visualization
- Compatible with multiple automation systems
- Published clone
- High sensitivity and specificity for squamous intraepithelial neoplasms in the cervix, anal canal, throat, and other sites of HPV infection
Flexible formats available:
|
Volume |
Cat. No. (AU) |
Cat. No. (NZ) |
|---|---|---|
|
0.1 mL concentrate |
CM416M14RUO |
CM416M14 |
|
0.5 mL concentrate |
CM416M15RUO |
CM416M15 |
|
1 mL concentrate |
CM416M16RUO |
CM416M16 |
|
1 mL predilute |
CM416M17RUO |
CM416M17 |
|
7 mL predilute |
CM416M18RUO |
CM416M18 |
|
25 mL predilute |
CM416M10RUO |
CM416M10 |
RUO version available in Australia.
IVD version available in New Zealand.
View full benefits of p16
Contact us for a quote
View Cell Marque latest IHC Guide